|
Importance of Understanding Electron Group Geometry The number and type of hybrid orbitals resulting from the electron group geometry have a direct impact on both electron group and molecular geometries.īy understanding this relationship, we can explain variations in bond angles observed within molecules. Impact on Electron Group and Molecular Geometries These hybrid orbitals are then used for bonding within molecules. Hybridization refers to the process where atomic orbitals mix to form new hybrid orbitals with different shapes and energies. 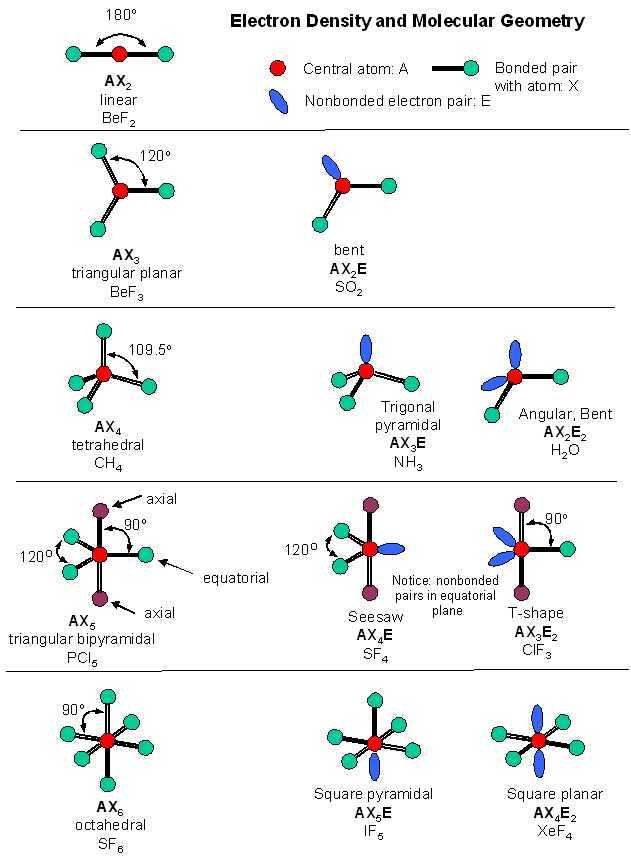
The electron group geometry influences the hybridization state of an atom. This, in turn, affects how atomic orbitals combine to form new hybrid orbitals involved in bonding. The electron group geometry plays a significant role in determining the hybridization state of an atom within a molecule. Understanding the electron group geometry is crucial. Electron Group Geometry in Molecular Shape and Hybridization Understanding these different molecular geometries helps chemists make predictions about how molecules will interact with each other and their physical properties such as polarity. Phosphorus pentachloride (PCl5) exhibits trigonal bipyramidal geometry. The bond angles vary depending on whether they are axial or equatorial positions. In this molecular geometry, there are five bonding pairs and no lone pairs around the central atom. Ammonia (NH3) is an example of a molecule with trigonal pyramidal geometry. The bond angle is slightly less than 109.5 degrees. This molecular geometry features three bonding pairs and one lone pair around the central atom. The bond angle is approximately 120 degrees.Īn example of a molecule with trigonal planar geometry is boron trifluoride (BF 3). In this molecular geometry, there are three bonding pairs and no lone pairs around the central atom. Let’s explore some common examples: Trigonal Planar It provides a framework for predicting the arrangement of bonded atoms and lone pairs around a central atom.ĭifferent combinations of bonded atoms and lone pairs result in various molecular geometries. The VSEPR (Valence Shell Electron Pair Repulsion) theory takes into account the number of valence electrons and the repulsion between them to determine molecular geometries. One commonly used theory to predict molecular shapes is the VSEPR theory. Molecular geometry is all about how atoms are arranged in a molecule, and it plays a crucial role in determining the physical properties of compounds.īy understanding molecular geometry, scientists can predict bond angles, polarity, and other important characteristics of molecules. Molecular Geometry in Introductory Chemistry They provide insights into how atoms arrange themselves to form stable compounds.īy knowing the electron and molecular geometries, scientists can predict various properties such as polarity, bond angles, and intermolecular forces. Importance in Understanding Chemical Bondingīoth electron and molecular geometries play crucial roles in understanding chemical bonding. Lone pairs can significantly impact the overall shape of a molecule, altering its properties and reactivity. These are pairs of electrons that do not participate in bonding but still occupy space around the central atom. Molecular geometry goes beyond electron geometry by taking into account lone pairs of electrons. This information is vital for understanding how different molecules interact and behave. Determining Molecular ShapeĮlectron geometry serves as a foundation for determining molecular shape through VSEPR (Valence Shell Electron Pair Repulsion) theory.īy considering the number of electron groups around a central atom, we can predict the overall shape of a molecule. While electron geometry focuses on the arrangement of electron groups, molecular geometry considers only the positions of atoms. 
Difference Between Electron Geometry and Molecular Geometry Focus on Arrangement and PositionsĮlectron geometry and molecular geometry are two concepts in chemistry that help us understand the shapes of molecules. Understanding electron geometry helps us understand complex molecular structures and their behavior. 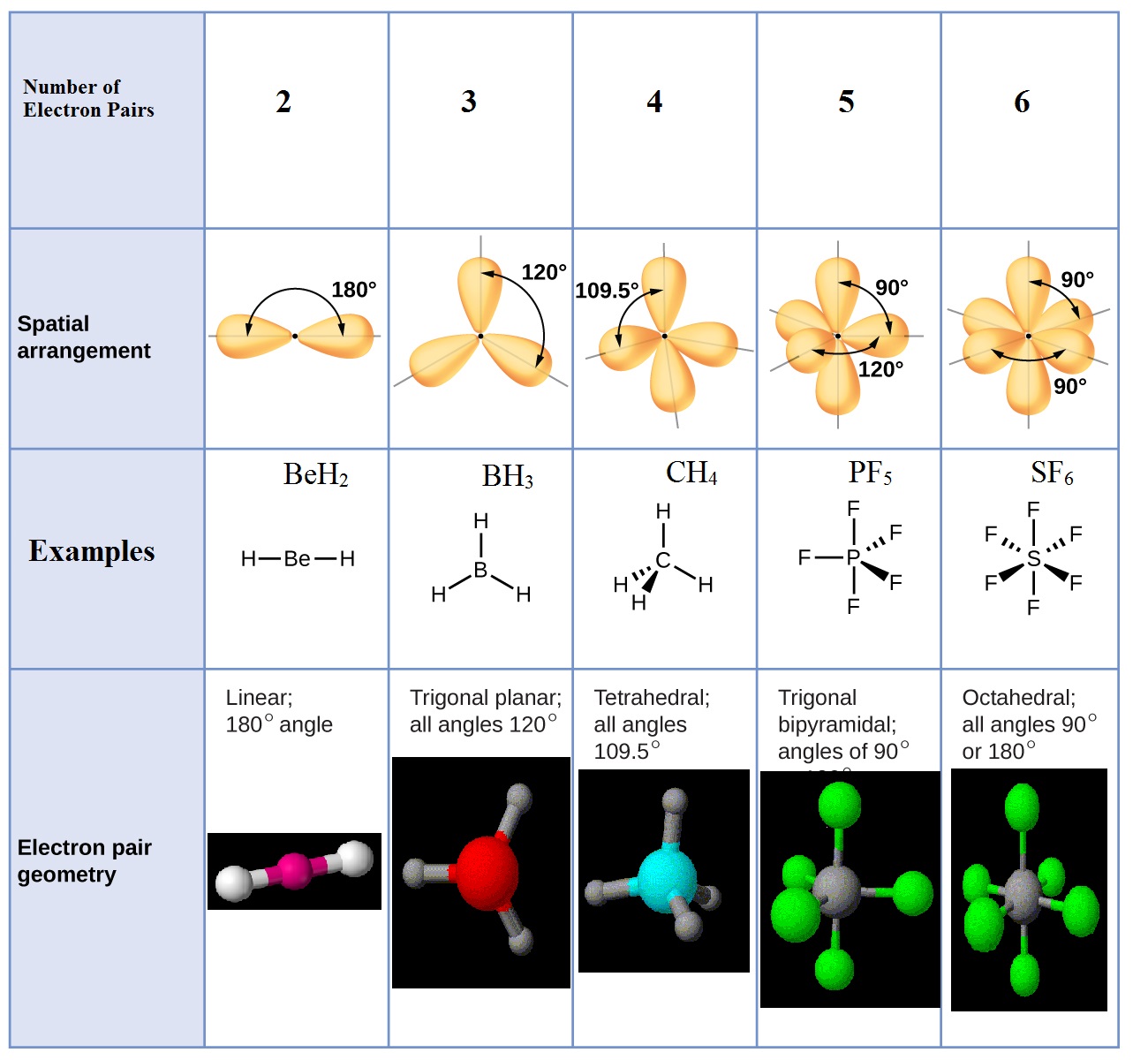 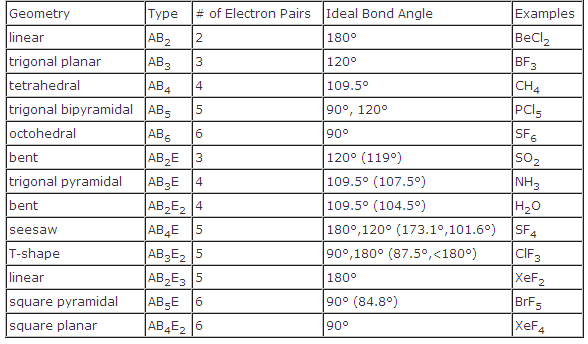
Techniques like Lewis dot structures and VSEPR help researchers find electron geometry by looking at valence electrons and the central atom. This concept is crucial in organic chemistry, where atom arrangement affects compound functionality. Different electron geometries lead to different molecular shapes, which affect stability and reactivity. Scientists use it to predict chemical behavior and reactions. Electron geometry is important for determining the shape and properties of molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
